Led by the Francis Crick Institute in the UK, the team brings together clinicians, scientists and patient advocates, with researchers spanning eight institutions across four countries. The award is funded jointly by Cancer Research UK and the National Cancer Institute in the US, two of the world’s largest cancer research funders, through the global Cancer Grand Challenges initiative. It marks the first time this prestigious international award has been granted to a Portuguese institution.
It is one of five new teams announced today, representing a total investment of $125m in research tackling some of the most complex challenges in cancer, with the aim of understanding the mechanisms that drive the disease. The initiative sends a strong signal about the importance of answering fundamental questions in biology as a foundation for future treatments.
Rethinking cancer as a whole-body disease
Cancer has traditionally been studied as a disease of cells and tissues. However, researchers are increasingly recognising that tumours do not exist in isolation. Nerves infiltrate many cancers, influencing tumour growth, the immune system and symptoms such as pain or weight loss. This is reshaping how cancer is understood – from a disease confined to tissues to one embedded in the biology of the whole organism.
What remains far less understood is how the brain itself perceives cancer. “At the heart of this project are two linked questions”, says Veiga-Fernandes. “First, can the brain sense that a tumour is growing in the body? And second, if it can, how does it respond – does it send signals that help suppress the cancer, or, in some cases, inadvertently support it?”.
The team’s name, InteroCANCEption, refers to the concept of interoception, the process by which the brain monitors the body’s internal state through signals carried by the nervous system. Every second, networks of sensory neurons relay information from organs to the brain, which in turn sends signals back to regulate bodily functions. The researchers believe that tumours may be part of this dialogue – and that understanding it could transform cancer biology.
To address this, the team will investigate how information about a tumour travels along neural pathways between organs and the brain, and how signals sent back from the brain may shape tumour progression and the immune response. This means looking beyond cancer cells themselves to the neural circuits and immune cells that surround them in the local environment.
“There is growing evidence that nerves can both promote and suppress tumour growth”, adds Veiga-Fernandes. “But we don’t yet understand the rules that govern these interactions. That is the gap we are trying to fill”.
Toward a new class of therapies
One of the most ambitious goals of the project is to explore whether manipulating neural activity could influence cancer outcomes. Electrical stimulation and small implanted devices are already used to treat neurological conditions such as Parkinson’s disease and other movement disorders. The team believes that similar implants – targeting specific regions of the spinal cord – could modulate how nerves communicate with tumours in organs such as the lung, pancreas and colon.
“If we can understand the circuits that connect the brain and tumours, we may be able to design entirely new kinds of therapies”, says María Martínez Lopez, postdoctoral researcher in the Veiga-Fernandes Lab. “Instead of targeting only cancer cells, we could target the signals that control how the body responds to cancer”.
In parallel, the researchers aim to identify key molecules involved in neuro-immune interactions, opening the possibility of new drug targets. “By deciphering these molecular signals, we may move beyond simply managing symptoms to addressing root causes. For example, we could harness a patient’s own immune cells outside the body to create tailored, vaccine-like treatments”, Lopez says.
While clinical applications remain a longer-term prospect, the work could lay the foundation for a new therapeutic field focused on neural modulation alongside molecular and immune-based strategies to better guide the nervous system’s activity in disease.
Patient perspectives at the roots of discovery
For Veiga-Fernandes, one aspect of the initiative stands out in particular: the involvement of patient advocates in the selection process. “Patients are part of the journey from the very beginning”, he says. “They even sit in on the interviews of shortlisted teams. It’s a powerful way of making the scientific process more open and bringing in the perspectives and experiences of the people who may ultimately benefit from what we discover”.
He also points out that the award underscores the value of basic science – research aimed at understanding how biology works at a fundamental level. “Many of the breakthroughs that transform medicine begin with curiosity-driven research”, he says. “This challenge is about asking deep questions. And history shows that answering those questions often leads to the most unexpected and transformative advances”.
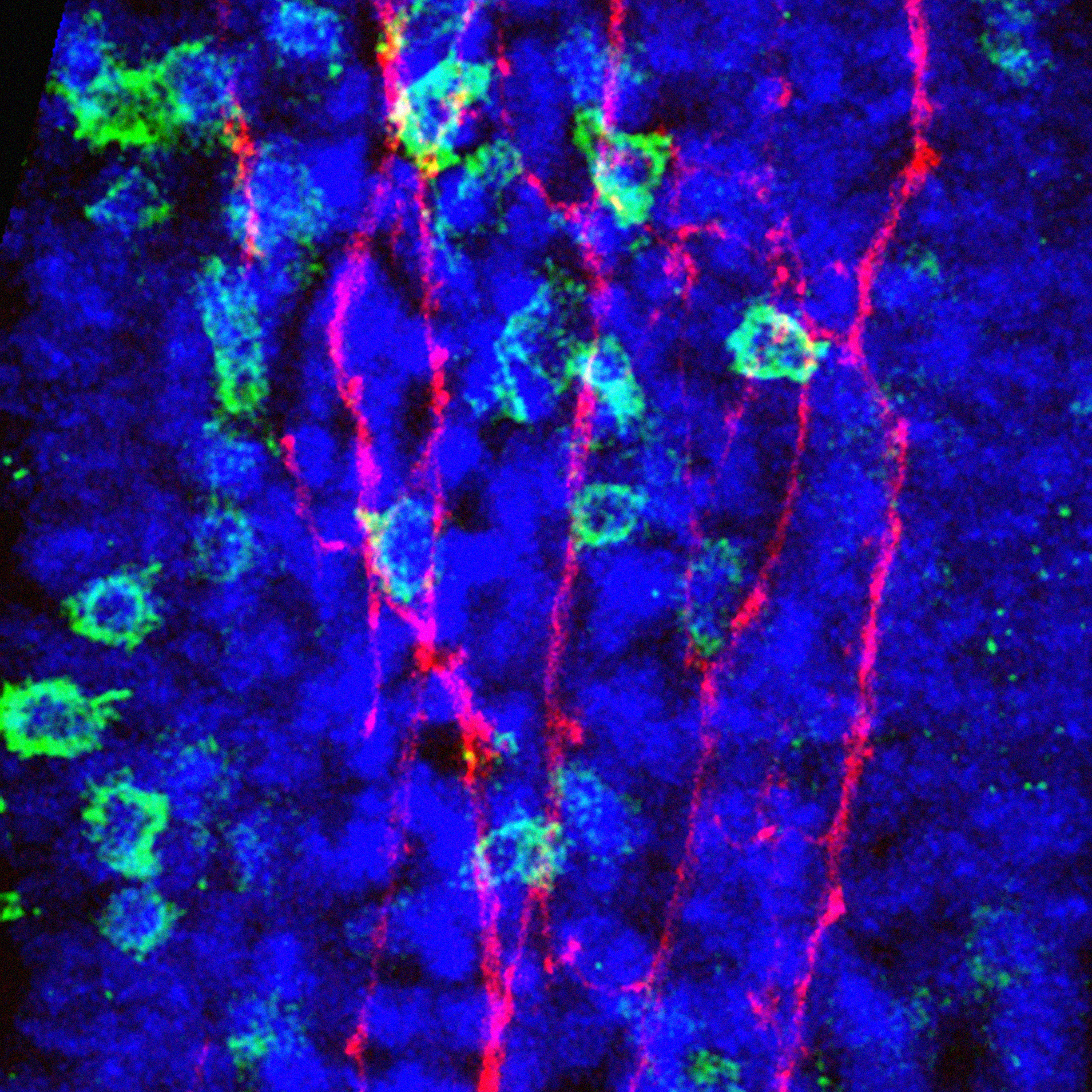
Image Caption: Fluorescence microscope image of tiny, finger-like projections in the gut showing nerve fibres (red) and immune cells (green) in close proximity (cell nuclei in blue). Understanding how nerve-immune interactions influence tumour growth is a central goal of the InteroCANCEption project.
Text by Hedi Young, Science Writer and Content Developer of the Champalimaud Foundation's Communication, Events & Outreach Team